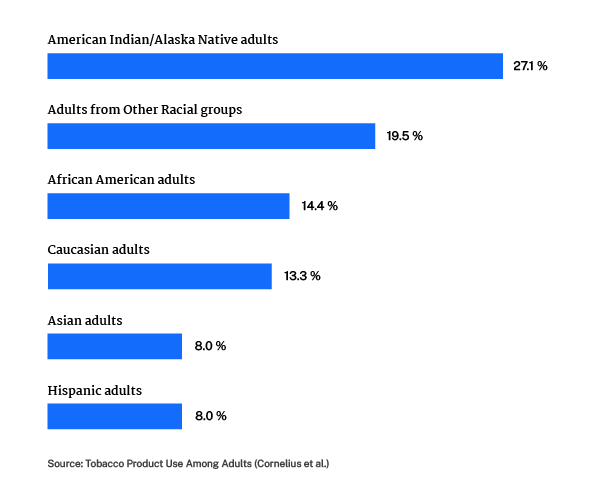FDA Must Prioritize Lowering Outsized Cancer Rates Caused by Cigarette Smoking
Evidence clearly demonstrates that vaping helps adult smokers quit, yet many communities are being misinformed about the relative risks of e-cigarettes.
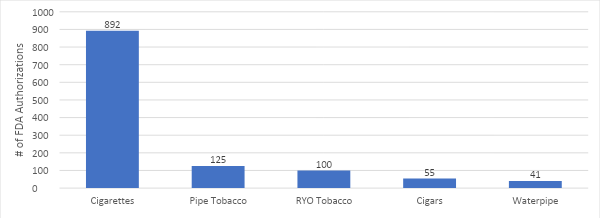
A policy of “harm reduction equity” that prioritizes eliminating cancer and other diseases caused by cigarette use should be the highest public health priority of the FDA. Yet FDA’s words and actions have led many to believe erroneously that vaping is as harmful as, or more harmful than smoking. FDA’s backward approach to harm reduction means that it is currently easier to get a new cigarette product approved than an e-cigarette alternative.
Smoking remains the number one health risk to adults in the United States. Approximately 34 million adults are still addicted to cigarettes – the only product which when used as intended will kill you. Nearly 1300 Americans die from a smoking related illness every day – almost 475,000 people annually. That is COVID scale event – every two years!
In the LBGTQ communities – the problem is worse. LGBTQ people out-smoke the general population by 68%. This high rate of LGBTQ smoking is a current and ongoing health crisis.
LGBTQ smokers need to receive accurate information from FDA about the relative risks of vaping and smoking – it is a matter of life and death.
LGBTQ communities experience health disparities because social stigma and discriminatory treatment often cause stress and anxiety. Common coping mechanisms include smoking tobacco, drugs and alcohol. Because of higher smoking rates, LGBTQ communities carry the burden of a much higher risk of cancer and other diseases:
- Smoking rates among LGB individuals in the US are higher than that of the heterosexual population (11.4% ) with 15.5% of LGBTQ adults smoking cigarettes. (CDC)
- In transgender communities , there are reports that 62% of adults smoke cigarettes. (Journal of Addiction Medicine)
- African Americans are more likely to die of smoking related illnesses than non-Hispanic Whites – causing around 45,000 deaths per year. (CDC).
- Smoking related deaths are on track to surpass HIV/AIDS as a cause of death among gay men. (NIH)
The FDA and the Center for Tobacco Policy (CTP) is charged, ultimately, with protecting all Americans from tobacco-related disease and death. This means the FDA must do everything to help people move off combustible tobacco and toward lower risk alternatives. FDA’s backward approach to harm reduction means that it is currently easier to get a new cigarette product approved than an e-cigarette alternative. And the fact that the FDA is on pace to eliminate more than 99% of vaping products from the market means that the only choice left remaining will be deadly cigarettes.
The Risk of Various Nicotine Products is Not the Same
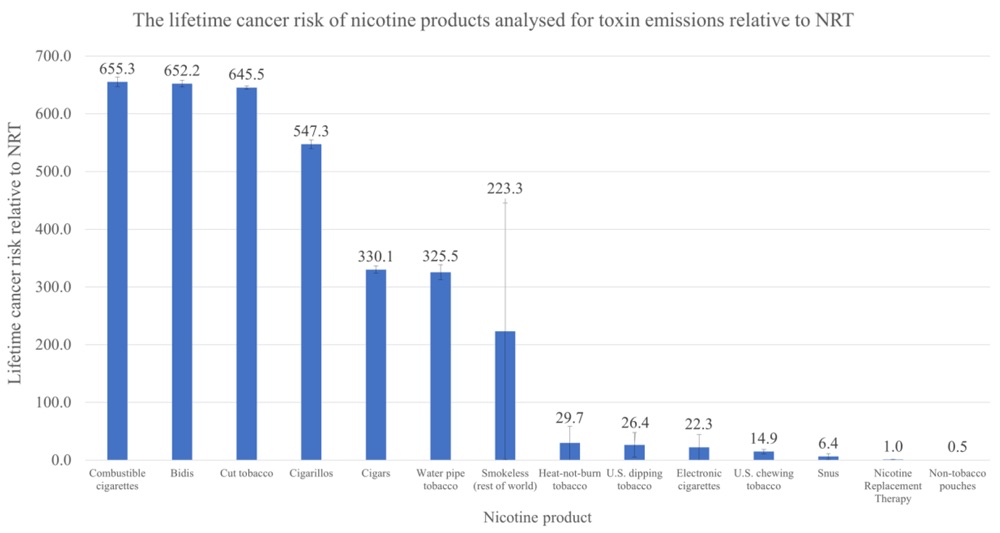
According to the American Cancer Society, cigarette smoking is the leading cause of cancer mortality in the United States – responsible for as much as 98% of all tobacco-related deaths. This is a generational health crisis that is preventable. Unfortunately it is also a problem that disparately impacts historically marginalized communities.
Regulators Must Tell the Truth
In a recent landmark essay in the American Journal of Public Health, 15 past presidents of the staunchly anti-tobacco Society for Research on Nicotine and Tobacco articulated the state of the science which demands an honest discussion about our public policy toward tobacco e-cigarettes. U.S.
policymakers are ignoring these scientific leaders’ warnings and missing the opportunity to reshape U.S. e-cigarette policy before more lives are lost.
According to these scientists, the need to pay attention to adult smokers is particularly important from a harm reduction equity perspective.
“African Americans suffer disproportionately from smoking-related deaths, a disparity that, a new clinical trial shows, vaping could reduce. Today’s smokers come disproportionately from lower education and income groups, the LGBTQ (lesbian, gay, bisexual, transgender, and queer or questioning) community, and populations suffering from mental health conditions
and from other drug addictions.”
Vaping is the Most Effective Tool to Deliver Harm Reduction Equity
While vaping is not risk free, we know that it is the most effective of all the nicotine alternatives. Since 2010, when e-cigarettes became widely available in the US, we have seen adults who smoke drop by a remarkable 62 percent. Other countries are far ahead of the U.S. in this regard.
Since 2015, the United Kingdom’s Health Agency Public Health England and the Royal College of Physicians each conducted a full review of all e-cigarette research and concluded that vapor products are at least 95% safer than combustible cigarettes. Now, Britain’s National Health Service promotes e-cigarettes as a means to quit smoking and even permits the sale of flavored vapor products inside hospitals.
And in April of 2023, the UK government announced its landmark program in which “One million smokers will be encouraged to swap cigarettes for vapes under a pioneering new ‘swap to stop’ scheme designed to improve the health of the nation and cut smoking rates.” The UK government is handing out free vaping starter kits to smokers to achieve its smoke free by 2030 goal..
Even in the U.S., the National Academies of Sciences Engineering and Medicine conducted its
own review of all the research and concluded in 2018:
- “There is conclusive evidence that completely substituting e-cigarettes for combustible tobacco cigarettes reduces users’ exposure to numerous toxicants and carcinogens present in combustible tobacco cigarettes.”
- “There is substantial evidence that completely switching from regular use of combustible tobacco cigarettes to e-cigarettes results in reduced short-term adverse health outcomes in
- several organ systems.”
- “The evidence about harm reduction suggests that across a range of studies and outcomes, e-cigarettes pose less risk to an individual than combustible tobacco cigarettes.”
So, while the FDA repeatedly talks about health equity, FDA’s refusal to acknowledge and apply the obvious principles of harm reduction to vaping dramatically highlights the inequity of its approach to harm reduction. Ultimately, the FDA will fail in its mission to end smoking and will particularly fail those in the LGBTQ communities who are disproportionately affected by smoking death and disease.
The Bottom Line
Less than 4% of underage youth indicate that they use vaping products “frequently.” FDA’s singular focus on decreasing this number by removing vaping products from the market is leaving millions of adult smokers with no alternatives to cigarettes. Vaping can be a benefit to public health and there is ample evidence supporting the potential of vaping to reduce smoking’s toll – particularly among the communities that remain addicted to cigarettes at higher rates. Making it harder for adults to access vapor products makes it harder for them quit and ensures that they engage in the riskiest behavior, (smoking) longer.
Given the unequal burden the LGBTQ community bears for cancer and other health risks directly associated with smoking, limiting adult choice for a lower risk product only serves to expand the harm reduction equity gap. Quitting smoking is hard. Only 10% of adults who try to quit will ultimately succeed. FDA needs to expand the understanding of less risky alternatives to smoking for the communities most at risk.